
Terence Williams Lab
Research Lab Overview
The research focus of the Terence Williams Lab is to elucidate the molecular biology of cancer vulnerability derived from genetic, epigenetic, metabolic and tumor micro-environmental alterations, and to find strategies targeting this vulnerability to improve cancer chemotherapy, radiotherapy, molecularly targeted therapy and immunotherapy.
The Williams Lab is translating its bench findings in the underlying mechanisms of the deregulation of genome surveillance systems and nutrient scavenging/caveolae-mediated endocytosis to clinic for the personalized diagnosis, prognosis and treatment of cancer patients.
The picture on the right shows Tiantian Cui, M.D., Ph.D., an assistant research professor in the Terence Williams Lab, sharing and discussing science at the 2022 conference of the American Association of Cancer Research (AACR) at New Orleans, LA.
Highlights
The overarching goal of our research is to understand the mechanisms of the inherent and acquired resistance of cancer cells to current available treatment modalities, particularly radiotherapy and chemotherapy at the genetic, transcriptomic and proteomic levels.
Such knowledge allows us to design and implement novel therapy strategies for poor-prognosis cancers, including gastrointestinal (pancreatic, esophago-gastric, colorectal and hepatobiliary), thoracic (non-small cell lung cancer), thyroid, and head and neck malignancies. We use various mouse models (transgenic, genetically engineered, patient-derived xenograft and orthotopic models) as well as patient tissues (tumor and normal) in combination with basic molecular and cellular techniques to address the fundamental cancer biological questions about how cancers initiate, develop and progress to aggressive, therapeutically-resistant and metastatic disease.
We identify and validate our biomarkers in clinically-relevant disease model systems for the diagnosis, treatment and prognosis of cancer patients. We collaborate with many other labs at City of Hope, across the U.S. and internationally in five main overlapping research areas with the goal of driving the development of new clinical trials:
- Caveolae-mediated endocytosis and nutrient scavenging
- Oncogene mediated radioresistance
- Identifying and targeting novel DNA repair and DNA damage response pathways modulating tumorigenesis and therapeutic response
- Developmental therapeutics (e.g., protein-drug conjugates, RNA, radiopharmaceuticals, radioimmunotherapy)
- Molecular profiling and predictive biomarkers in immuno-oncology
In the News

Congratulations to Dr. Sergio Corrales-Guerrero on his Commencement at the Ohio State University on December 18, 2023. We wish him all the best in his new position!
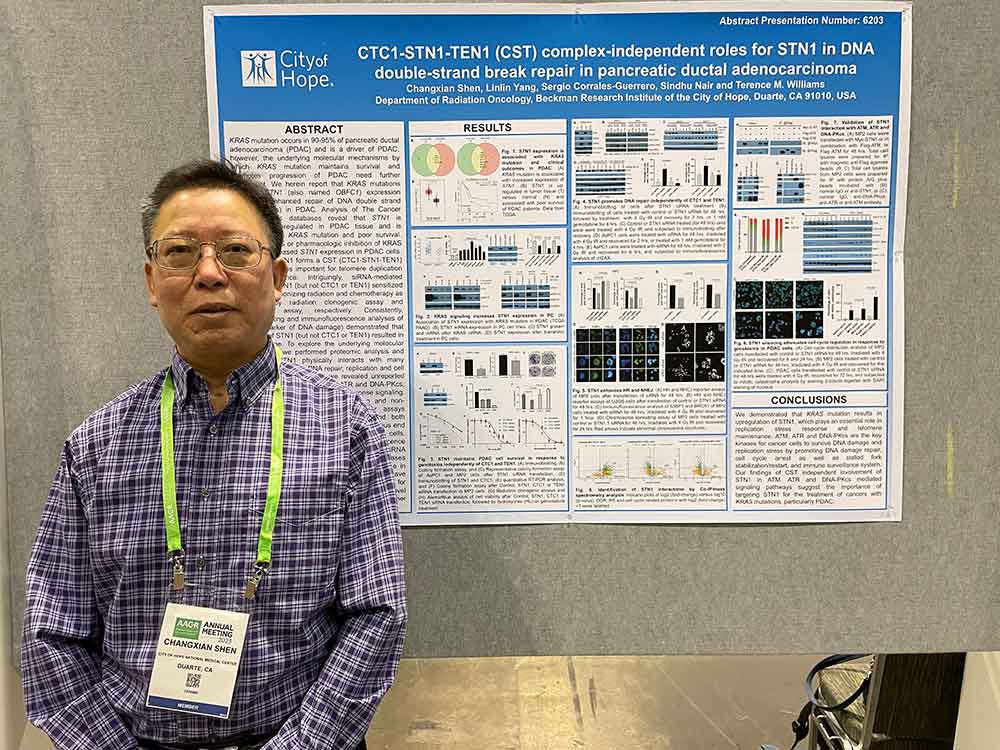
Dr. Changxian Shen stands next to his poster, “CTC1-STN1-TEN1 (CST) complex-independent roles for STN1 in DNA double-strand break repair in pancreatic ductal adenocarcinoma,” at the 2023 conference of the American Association of Cancer Research (AACR) in Orlando, Florida.
STN1 forms a CST (CTC1-STN1-TEN1) complex crucial for telomere duplication and maintenance. We found that depletion of STN1 but not CTC1 or TEN1 sensitizes pancreatic ductal adenocarcinoma cells to ionizing radiation and chemotherapy drugs. Proteomic analysis revealed that STN1 physically interacts with many proteins crucial for general DNA repair, DNA replication and cell cycle progression.
Our findings have revealed novel CST complex independent mechanisms for STN1 in DNA repair and suggested STN1 is a promising biomarker for management of cancer patients with pancreatic ductal adenocarcinoma.
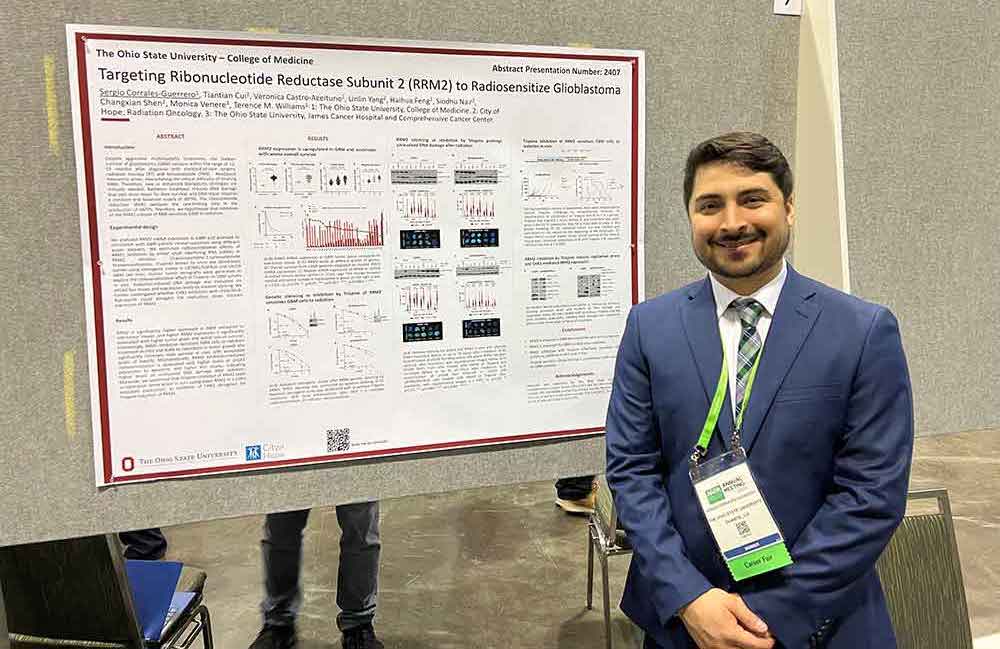
Sergio Corrales-Guerrero stands next to his poster “Targeting ribonucleotide reductase subunit 2 (RRM2) to radio-sensitize glioblastoma” at the 2023 conference of the American Association of Cancer Research (AACR) in Orlando, Florida.
Glioblastoma (GBM) is an aggressive malignant primary brain tumor that is resistant to currently available treatment modules. Intrinsic and acquired resistance to radiotherapy largely contributes to the poor clinical outcomes of GBM patients after systemic treatment. We discovered that ribonucleotide reductase subunit 2 (RRM2) upregulation promotes radiation resistance and selectively targeting RRM2 with blood-brain barrier permeable Triapine sensitizes GBM to radiotherapy.
Intriguingly, targeting RRM2 with Triapine induces synthetic lethality of GBM cells with CHK1 inhibitor Rabusertib. Our findings suggest that combining Triapine with radiation or Rabusertib may improve the therapeutic outcome of GBM patients.
Dr. Linlin Yang stands next to her poster "Targeting KRAS mutant non-small cell lung cancer with EGFR aptamer displaying 3WJpRNA nanoparticles" at the 2022 conference of the American Association of Cancer Research (AACR) at NOLA
In this study, we assembled novel EGFRaptmer-3WJ-siKRAS mutation-targeted nanoparticles to target EGFR-expressing human NSCLC harboring a KRASG12C mutation to silence KRASG12C expression in a tumor-specific fashion. We found that EGFRapt-3WJ-siKRASG12C nanoparticles potently and selectively depleted cellular KRASG12C expression, resulting in attenuation of downstream MAPK pathway signaling, cell proliferation, migration/invasion ability, sensitization of NSCLC cells to chemoradiotherapy, and in vivo growth inhibition of KRASG12C NSCLC tumor xenografts. Together, this study suggests that the 3WJ pRNA-based platform has the potential to suppress mutant KRAS activity for the treatment of KRAS-driven human cancers, and warrants further development for clinical translation.
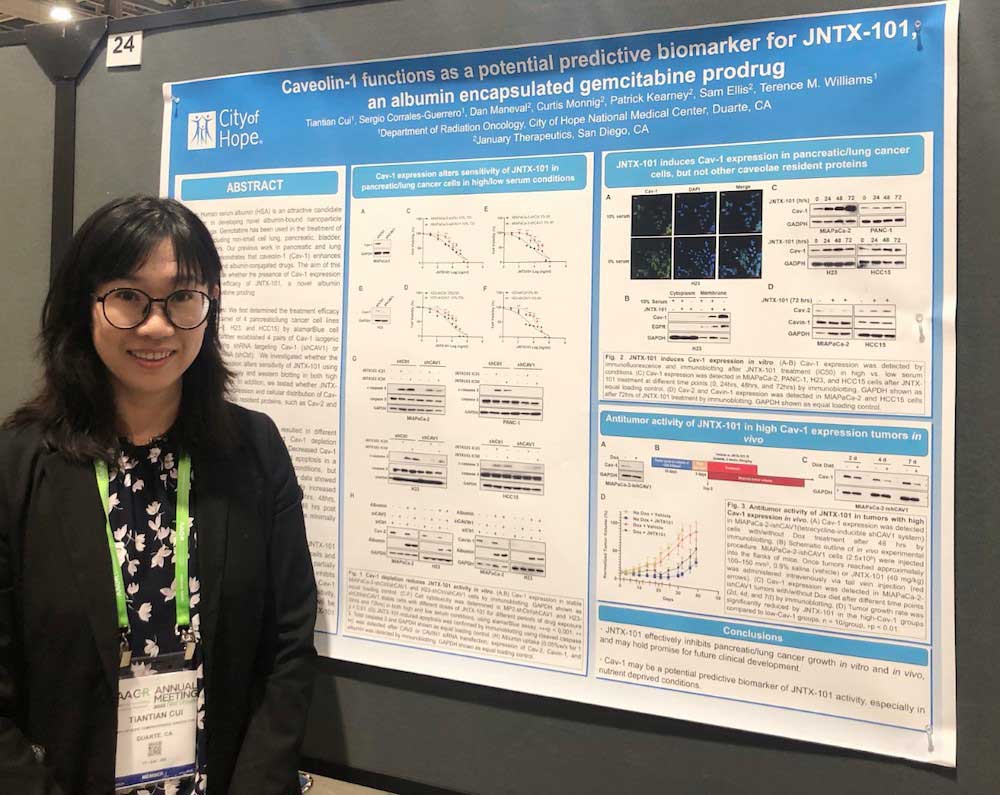
Dr. Tiantian Cui’s poster ‘Caveolin-1 functions as a potential predictive biomarker for JNTX-101, an albumin encapsulated gemcitabine prodrug’ at the 2022 conference of the American Association of Cancer Research (AACR) at NOLA
JNTX-101, an albumin-encapsulated gemcitabine prodrug, effectively inhibits pancreatic and lung cancer tumor growth and may hold promise for future clinical development. Cav-1 expression dictates sensitivity to JNTX-101, at least partially through reduced apoptotic cell death. Cav-1 may be a potential predictive biomarker of JNTX-101 efficacy, especially in nutrient-deprived conditions.
COH Shared Resources Grant
Dr. Terence Williams and Dr. Tiantian Cui have received a COH Shared Resources Grant in 2021 entitled “Targeting Tumor Endocytosis to Drive Novel Anti-tumor Strategies”. This study will identify individual biomarkers or biomarker signatures that predict for efficacy of albumin uptake and develop pharmacological strategies to increase albumin uptake prior to delivery of novel albumin-conjugated cytotoxic chemotherapeutics (as well as the FDA-approved nab-paclitaxel) or targeted agents, leading to rapid clinical translation of novel and innovative oncologic biomarkers and treatments.
Career Opportunities
The goal of Dr. Williams’ Lab is to revolutionize the management of cancer patients. To help achieve this goal, we are working to develop an innovative and dynamic scientific research team at City of Hope.
To accomplish our mission, we promote an environment centered on teamwork and learning for all team members at every stage of training (staff scientist level, post-doctoral, graduate and undergraduate).
Specifically, we are looking for enthusiastic, ambitious and self-motivated individuals who thrive in a fast-paced, competitive and challenging research environment and who can interact well with others as members of our research family. If you work hard and have a dream to make a significant difference in the health care of cancer patients, we invite you to contact us. Inquiries can be directed to Phyllis Burch at pburch@coh.org.
Please include the following:
- Short cover letter
- CV, biosketch, or resume with names of references
- Areas of interest in our lab
Mailing address:
Department of Radiation Oncology
City of Hope National Medical Center
Beckman Research Institute
1500 East Duarte Road
Duarte, CA 91010
Inquiries can be directed to:
- Phyllis Burch
- Email: pburch@coh.org
Dr. Williams is a radiation oncologist.



Dr. Tiantian Cui, M.D., Ph.D., graduated from Binzhou Medical University in 2005. In 2008, she attained her master’s degree and completed her residency in the Radiation Oncology Department at Sun Yat-sen University Cancer Center. With a Ph.D. scholarship from the Interdisciplinary Center for Clinical Research (IZKF, Germany), Dr. Cui joined Professor Iver Petersen's group at Fiedrich-Schiller University.
She earned her Ph.D. summa cum laude in 2011, working on the role of desmosome membrane proteins in human cancers. Her expertise lies in understanding molecular mechanisms of cancer initiation, progression and therapeutic resistance. Dr. Cui’s long research goal is to improve personalized cancer treatment by developing innovative and targeted strategies that result in better patient outcomes.


Changxian Shen, Ph.D., completed his B.S. at Hunan Normal University in Changsha, China, and his M.S. at Beijing Institute of Microbiology & Epidemiology in Beijing, China. After that, Shen moved to Germany, where he completed his Ph.D. in the field of molecular cancer biology at the University of Ulm. He finished his postdoctoral training at St. Jude Children’s Research Hospital in Memphis, Tennessee. Shen followed up with his studies in cancer biology and translational cancer research at Nationwide Children’s Hospital and Ohio State University in Columbus, Ohio.
Shen’s research has been focused on the roles for cell growth signaling pathways (including RAS-MAPK and PI3K-mTOR) in the regulation of cell cycle progression and DNA damage repair. His long-term goal is to elucidate the mechanisms of tumorigenesis and find better therapies for aggressive and hard-to-treat tumors, such as pancreatic and lung cancers, by exploring the genetic and epigenetic alterations of cancer cells.


Veronica Castro-Aceituno, Ph.D., R.D.N., graduated from San Carlos de Guatemala University and became a registered dietitian nutritionist in 2013. With a scholarship from the National Secretariat of Science and Technology of Guatemala, she joined Professor Deok-Chun Yang's laboratory. In 2018, she attained her Ph.D. by investigating the anti-cancer mechanism of various nanoparticles derived from oriental medicinal plants at Kyung Hee University in South Korea. Later, she joined Professor Young-Han Song's team at Hallym University as a postdoctoral researcher.
In 2019, Castro-Aceituno attained a grant from the National Research Foundation of South Korea to study the mechanism of activation by tetramerization of Drosophila p53 (Dp53) and the identification of the physiological function of the isoforms of Dp53. She joined the lab of Dr. Terence Williams at City of Hope in July 2021.
Her experience lies in the elucidation of the possible anticancer mechanism of new therapies for the treatment of cancer. Castro-Aceituno's research goals are to study tumor metabolism and cancer nutrient scavenging and to improve the quality of life of cancer of patients by shedding light on interdisciplinary methods that will avoid resistance to treatment and reduce its side effects, including cancer cachexia.
Research Focus
- Caveolae Mediated Endocytosis in Nutrient Scavenging and Tumor Metabolism/Progression
- Cav-1/Caveolae in Tumor Cachexia and Macropinocytosis
Education
- 2013-2018, Ph.D., Oriental Medicine Materials and Processing Department, Kyung Hee University, Suwon, South Korea
- 2005-2011, B.S., R.D.N., Nutrition School, Department of Chemical Science and Pharmacy, San Carlos University, Guatemala
Internship
- 2011, Intern, Clinical Nutrition, Hospital General San Juan de Dios, Guatemala
- 2011, Intern, Community Nutrition, Save the Children, Quiche, Guatemala
Professional Experience
- 2021-Present, Postdoctoral Research Fellow, Radiation Oncology Department, City of Hope National Medical Center, CA, USA
- 2018-2021, Postdoctoral Research Fellow, Molecular Oncology Laboratory, Ilsong Institute of Life Science, Hallym University, South Korea
- 2013-2018, Graduate Researcher, Oriental Medicine and Biotechnology Department, Hanbanbio Laboratory, Kyung Hee University, South Korea
- 2012-2013, Head of Nutrition Department (RDN), Hospital National de Melchor de Mencos Peten, Guatemala
Grants
- 2019-2021, Academic Balanced Development Support Grant. Nacional Research Foundation (NRF), South Korea. Project: “Mechanism of activation by tetramerization and identification of physiological function of Drosophila p53 isoforms”
Awards
- 2017, Best Oral Presentation Award, The Korean Society of Medicinal Crop Science, Journal of Korean Medicinal Crop Science Conference
- 2017, Korean Symposium on Medicinal Crop Science and Autumn Symposium, South Korea
- 2017, Internationalization Scholarship, Graduate studies, Kyung Hee University, South Korea Period, Spring 2016 – Spring 2017
- 2016, Paper Publication in Journal, Award-Scholarship, Kyung Hee University, South Korea, Fall 2016, Paper: “In Vitro Cellular & Developmental Biology-Animal, 2016 52(10): 1012-1019”
- 2013, Scholarship for International Students. Graduate studies. Kyung Hee University, South Korea, Period: Fall 2013 – Spring 2016
- 2013, President Scholarship, Graduate studies, Kyung Hee University, South Korea, Period: Fall 2013 – Spring 2017
Memberships
- 2018, Organization for Women in Science for the Developing World
- 2018, International Network of Science, Technology and Innovation of Guatemala
Publications
- Yi, Myoung H., Shakina Yesmin Simu, Sungeun Ahn, Verónica Castro Aceituno, et al. Antiobesity Effect of Gold Nanoparticles from Dendropanax morbifera Léveille by Suppression of Triglyceride Synthesis and Downregulation of PPARγ and CEBPα Signaling Pathways in 3T3-L1 Mature Adipocytes and HepG2 Cells. Current Nanoscience 2020;16(2): 196-203.
- Park, Ji-Hong, Tram Thi Ngoc Nguyen, Eun-Mi Lee, Veronica Castro-Aceituno, et al. Role of p53 isoforms in the DNA damage response during Drosophila oogenesis. Scientific reports 2019; 9(1): 1-12.
- Kim, Y.J., Perumalsamy, H., Markus, J., Balusamy, S.R., Wang, C., Ho Kang, S., Lee, S., Park, S.Y., Kim, S., Castro-Aceituno, V. and Kim, S.H. Development of Lactobacillus kimchicus DCY51Tmediated gold nanoparticles for delivery of ginsenoside compound K: in vitro photothermal effects and apoptosis detection in cancer cells. Artificial cells, nanomedicine, and biotechnology 2019; 47(1): 30- 44.
- Shim, Yeon Jae, Veronika Soshnikova, Gokulanathan Anandapadmanaban, Ramya Mathiyalagan, Zuly Elizabeth Jimenez Perez, Josua Markus, Yeon Ju Kim, Verónica CastroAceituno, and Deok Chun Yang. Zinc Oxide Nanoparticles Synthesized by Suaeda japonica Makino and Their Photocatalytic Degradation of Methylene Blue. Optik 2019; 182: 1015-1020.
- Simu, S. Y., Ahn, S., Castro-Aceituno, V., et al. Gold Nanoparticles Synthesized with Fresh Panax ginseng Leaf Extract Suppress Adipogenesis by Downregulating PPARγ/CEBPα Signaling in 3T3-L1 Mature Adipocytes. Journal of Nanoscience and Nanotechnology 2019; 19(2): 701-708
- Mathiyalagan, R., Wang, C., Kim, Y. J., Castro-Aceituno, V., et al. Preparation of Polyethylene Glycol-Ginsenoside Rh1 and Rh2 Conjugates and Their Efficacy against Lung Cancer and Inflammation. Molecules 2019; 24(23): 4367.
- Kim, Y. J., Perumalsamy, H., Castro-Aceituno, V., et al. Photoluminescent And SelfAssembled Hyaluronic Acid-Zinc Oxide-Ginsenoside Rh2 Nanoparticles And Their Potential Caspase-9 Apoptotic Mechanism Towards Cancer Cell Lines. International Journal of Nanomedicine 2019; 14: 8195.
- Kim, C.G.*, Castro-Aceituno, V.*, et al. Caspase-3/MAPK pathways as main regulators of the apoptotic effect of the phyto-mediated synthesized silver nanoparticle from dried stem of Eleutherococcus senticosus in human cancer cells. Biomedicine & Pharmacotherapy 2018; 99: 128-133.
- Simu, S. Y*., Castro‐Aceituno, V*., et al. Fermentation of soybean hull by Monascus pilosus and elucidation of its related molecular mechanism involved in the inhibition of lipid accumulation. An in sílico and in vitro approach. Journal of Food Biochemistry 2018; 42(1): e12442.
- Singh, P., Ahn, S., Kang, J.P., Veronika, S., Huo, Y., Singh, H., Chokkaligam, M., El-Agamy Farh, M., Aceituno, V.C., et al. In vitro anti-inflammatory activity of spherical silver nanoparticles and monodisperse hexagonal gold nanoparticles by fruit extract of Prunus serrulata: a green synthetic approach. Artificial cells, nanomedicine, and biotechnology 2018; 46(8): 2022-2032.
- Lee, H.A., Castro-Aceituno, V., et al. Rhizome of Anemarrhena asphodeloides as mediators of the eco-friendly synthesis of silver and gold spherical, face-centred cubic nanocrystals and its antimigratory and cytotoxic potential in normal and cancer cell lines. Artificial cells, nanomedicine, and biotechnology 2018; 46(sup2): 285-294.
- Oh, K.H., Soshnikova, V., Markus, J., Kim, Y.J., Lee, S.C., Singh, P., Castro-Aceituno, V., et al. Biosynthesized gold and silver nanoparticles by aqueous fruit extract of Chaenomeles sinensis and screening of their biomedical activities. Artificial Cells, Nanomedicine, and Biotechnology 2018; 46(3): 599- 606.
- Huo, Y., Singh, P., Kim, Y.J., Soshnikova, V., Kang, J., Markus, J., Ahn, S., Castro-Aceituno, V., et al. Biological synthesis of gold and silver chloride nanoparticles by Glycyrrhiza uralensis and in vitro applications. Artificial Cells, Nanomedicine, and Biotechnology 2018; 46(2): 303-312.
- Ahn, S., Singh, P., Jang, M., Kim, Y. J., Castro-Aceituno, V., et al. Gold nanoflowers synthesized using Acanthopanacis cortex extract inhibit inflammatory mediators in LPS-induced RAW264. 7 macrophages via NF-κB and AP-1 pathways. Colloids and Surfaces B: Biointerfaces 2018; 62, 398-404.
- Soshnikova, V., Kim, Y.J., Singh, P., Huo, Y., Markus, J., Ahn, S., Castro-Aceituno, V., Kang, J., Chokkalingam, M., Mathiyalagan, R. and Yang, D.C., 2017. Cardamom fruits as a green resource for facile synthesis of gold and silver nanoparticles and their biological applications. Artificial Cells, Nanomedicine, and Biotechnology 2018; 41(1): 108-117.
- Castro-Aceituno, V*., Abbai, R*., et al. Pleuropterus multiflorus (Hasuo) mediated straightforward eco-friendly synthesis of silver, gold nanoparticles and evaluation of their anti-cancer activity on A549 lung cancer cell line. Biomedicine & Pharmacotherapy 2017; 93: 995-1003
- Wang, Dandan, Josua Markus, Chao Wang, Yeon-Ju Kim, Ramya Mathiyalagan, Verónica Castro Aceituno, et al. Green synthesis of gold and silver nanoparticles using aqueous extract of Cibotium barometz root. Artificial cells, nanomedicine, and biotechnology 2017; 45(8): 1548-1555.
- Singh, P., Singh, H., Ahn, S., Castro-Aceituno, V., et al. Pharmacological importance, characterization and applications of gold and silver nanoparticles synthesized by Panax ginseng fresh leaves. Artificial cells, nanomedicine, and biotechnology 2017; 45(7): 1415-1424.
- Park, J. K., V. Castro-Aceituno, et al. "Ginsenoside from Panax ginseng Meyer Enhances the Cytotoxic and Apoptotic Effect of Cisplatin in A549 Human Lung Cancer Cells." Indian Journal of Pharmaceutical Sciences 2017; 79.3: 468-474.
- Hurh, J., Markus, J., Kim, Y. J., Ahn, S., Castro-Aceituno, V., et al. Facile reduction and stabilization of ginsenoside-functionalized gold nanoparticles: optimization, characterization, and in vitro cytotoxicity studies. Journal of Nanoparticle Research 2017; 19(9): 313.


Sergio Corrales-Guerrero, M.S., graduated from the University of Navarra in Spain with a bachelor’s degree in biology in 2014. He completed his M.S. degree in cancer research and molecular biomedicine in 2015 at the University of Manchester in the U.K. Between 2016 and 2018, he worked in the Epidemiology and Biostatistics Department at Memorial Sloan Kettering Cancer Center. Then, he moved on to the Biomedical Sciences Graduate Program for his Ph.D. studies at Ohio State University, joining the lab of Dr. Terence Williams in 2019. Now based at City of Hope, he focuses on understanding the molecular mechanisms underlying the enhanced and uncontrolled growth and survival of cancer cells, with an aim to develop novel treatment approaches to improve cancer patient outcomes.
Research Focus
- Caveolin-1 mediated nutrient uptake in tumorigenesis and drug resistance
- Glioblastoma radioresistance
Education
- 2018-present, Ph.D. Graduate Student, The Ohio State University College of Medicine, Columbus, Ohio, U.S.
- 2014-2015, M.S. Graduate Student, Cancer Research and Molecular Biomedicine, The University of Manchester, Manchester, U.K.
- 2010-2014, B.S., Biology, University of Navarra (Universidad de Navarra), Pamplona, Spain
Internship
- 2021-present, City of Hope, Duarte, California, U.S.
- 2012, Genetics Institute, National University of Colombia (Universidad Nacional de Colombia), Bogotá, Colombia
Professional Experience
- 2019-present, Graduate Research Assistant, The Ohio State University College of Medicine, Columbus, Ohio, U.S.
- 2016-2018, Senior/Research Technician, Memorial Sloan Kettering Cancer Center, New York, New York, U.S.
Awards
- 2020, Minority Scholar in Cancer Research Award, American Association for Cancer Research
- 2018, University Fellowship, The Ohio State University
- 2010-2012, Alumni Scholarship for Undergraduate Studies, University of Navarra
Memberships
- 2020-present, American Association for Cancer Research
Publications
- Robb R, Kuo JC, Liu Y, Corrales-Guerrero S, et al. A novel protein-drug conjugate, SSH20, demonstrates significant efficacy in caveolin-1-expressing tumors. Mol Ther Oncolytics 2021; 22:555-564. doi: 10.1016/j.omto.2021.07.013.
- Benej M, Wu J, Kreamer M, Kery M, Corrales-Guerrero S, Papandreou I, Williams TM, et al. Pharmacological Regulation of Tumor Hypoxia in Model Murine Tumors and Spontaneous Canine Tumors. Cancers 2021 Apr 3th. doi: 10.3390/cancers13071696.
- Orlow I, Shi Y, Kanetsky PA, Thomas NE, Luo L, Corrales-Guerrero S, et al. The interaction between Vitamin D Receptor polymorphisms and sun exposure around time of diagnosis influences melanoma survival. Pigment Cell Melanoma Res. 2017 Oct 9th. doi: 10.1111/pcmr.12653.


Haihua Feng, Ph.D., graduated from China Pharmaceutical University in 2010, and got his Ph.D. degree in the Department of Pathophysiology at Nanjing Medical University in 2015. After that, he did his postdoctoral research training at Albany Medical Center and Cleveland Clinic in the DNA damage and DNA replication field.
Dr. Feng joined the lab of Terence Williams, M.D., Ph.D., in 2021 to continue translating his research from the bench to the clinic. Dr. Feng’s long-term research goal is to identify the molecular mechanisms of DNA damage response and DNA replication and develop novel strategies to sensitize tumors to radiotherapy and chemotherapy.
Research Focus
- DNA damage response and DNA replication stress response
- Strategies to sensitize tumors to chemoradiation
Education
- 2010-2015, Ph.D., Department of Pathophysiology, Nanjing Medical University, Nanjing, China
- 2012-2014, Exchange student, Molecular and Cellular Physiology, Albany Medical College, New York, New York, U.S.
- 2005-2010, B.S., Pharmaceutical Engineering, China Pharmaceutical University, Nanjing, China
Professional Experience
- 2021-present, Postdoctoral Research Fellow, Department of Radiation Oncology, City of Hope, Los Angeles, California, U.S.
- 2018- 2021, Postdoctoral Research Fellow, Department of Cancer Biology, Cleveland Clinic, Cleveland, Ohio, U.S.
- 2017-2018, Postdoctoral Research Fellow, Department of Molecular and Cellular Biology, Albany Medical College, New York, New York, U.S.
- 2016-2017, Research Associate at Jiangnan University, and Eyecure company, Wuxi, China
Publications
- Feng H, Lu J, Song X, Thongkum A, Zhang F, Lou L, Reizes O, Almasan A, Gong Z. CK2 kinase-mediated PHF8 phosphorylation controls TopBP1 stability to regulate DNA replication. Nucleic Acids Res. 2021;49(4):2400-2401.
- Shen C, He Y, Chen Q, Feng H, Williams TW, Lu Y, He Z. Narrative review of emerging roles for AKT-mTOR signaling in cancer radioimmunotherapy. Ann Transl Med 2021;9(20):1596.
- Xie L, Feng H, Li S, Meng G, Liu S, Tang X, Ma Y, Han Y, Xiao Y, Gu Y, Shao Y, Park C-M, Xian M, Huang Y, Ferro A, Wang R, Moore PK, Wang H, Ji Y. SIRT3 Mediates the Antioxidant Effect of Hydrogen Sulfide in Endothelial Cells. Antioxidants & Redox Signaling. 2016; 24(6): 329-343.
- McMahon JJ, Yu W, Yang J, Feng H, Helm M, McMahon E, Zhu X, Shin D, Huang Y. Seizure-dependent mTOR activation in 5-HT neurons promotes autism-like behaviors in mice. Neurobiol Dis. 2015; 73: 296–306.
- Qichun Zhang Q, Yun Zhang Y, Haihua Feng H, Rui Guo, Lai Jin, Rong Wan, Lina Wang, Cheng Chen, and Shengnan Li. High Density Lipoprotein (HDL) Promotes Glucose Uptake in Adipocytes and Glycogen Synthesis in Muscle Cells. PLoS One. 2011; 6(8): e23556.


Sindhu Nair, D.M.D., Ph.D., graduated with a bachelor’s degree in dental surgery (B.D.S.) in 2010 and then completed her D.M.D. residency (master's degree in oral and maxillofacial pathology) in 2014 from Rajiv Gandhi University of Health Sciences, India.
Her master's thesis research involved understanding field cancerization and neoangiogenesis in oral squamous cell carcinoma and its correlation with clinicopathological parameters. In 2021, she received her Ph.D. in cancer biology from the University of Alabama at Birmingham, where her dissertation research involved investigating dysregulated alternative splicing of ANXA7 and its functional impact on receptor tyrosine kinase signaling in glioblastoma.
Additionally, she characterized EGFR ectodomain mutations giving rise to and sustaining cetuximab resistance in head and neck cancer. She joined Dr. Williams’ lab in 2021 as a postdoctoral fellow. Dr. Nair’s expertise lies in histopathology, molecular mechanisms of signal transduction, endocytic trafficking and mutational causes of therapeutic resistance. Her long-term goal is to merge new technologies with basic cancer research in order to develop personalized and efficient therapies for cancer.
Research Focus
- Targeted CRISPR screening for novel biomarkers mediating chemoradioresistance
- Endocytic trafficking of nanotherapeutics in pancreatic and lung cancer
Appointment
Postdoctoral fellow, Department of Radiation Oncology, City of Hope, Duarte, CA
Education
- 2005-2010, Bachelor of Dental Surgery, Rajiv Gandhi University of Health Sciences, India
- 2011-2014, Master’s in Dental Sciences (Oral Pathology), Rajiv Gandhi University of Health Sciences, India
- 2016-2021, Ph.D. (Cancer Biology), The University of Alabama at Birmingham, Birmingham, AL, U.S.
Professional Experience
- 2021-present, Postdoctoral fellow, Department of Radiation Oncology, City of Hope, Los Angeles, CA, U.S.
- 2015-2016, Assistant Professor, Department of Oral & Maxillofacial Pathology, AJ Institute of Dental Sciences, India
Awards
- 2017, Intramural pilot grant award, Department of Radiation Oncology, The University of Alabama at Birmingham, Birmingham, AL, U.S.
- 2018, Graduate Student Government Travel Grant Award. The University of Alabama at Birmingham, Birmingham, AL, U.S.
Memberships
- Society for Neuro-Oncology
- American Association of Cancer Research
- Indian Dental Association
- Indian Association of Oral & Maxillofacial Pathologists
Publications
- Nair S, Trummell HQ, Rajbhandari R, Thudi NK, Nozell SE, Warram JM, Willey CD, Yang ES, Placzek WJ, Bonner JA, Bredel M. Novel EGFR ectodomain mutations associated with ligand-independent activation and cetuximab resistance in head and neck cancer. PLOS ONE 2020; 15(2): e0229077
- Meares GP, Rajbhandari R, Gerigk M, Tien CL, Chang C, Fehling SC, Rowse A, Mulhern KC, Nair S, Gray GK, Berbari NF, Bredel M, Benveniste EN, Nozell SE. MicroRNA-31 is required for astrocyte specification. Glia 2018; 66(5):987-998
- Nair S, Kotrashetti V, Nayak R, Hosmani J. Occlusal groove patterns and cusp number in permanent mandibular first and second molar among Indian population - A pilot study. J Advanced Clinical & Research Insights 2018; 5(4):115-118
- Nair S, Nayak R, Bhat K, Kotrashetti V, Babji D.
Immunohistochemical expression of CD105 and TGF-β1 in oral squamous cell carcinoma and adjacent apparently normal oral mucosa and its correlation with clinicopathological features. Appl Immunohistochem Mol Morphol. 2016; 24(1):35-41. - Nair S, Kotrashetti V, Nayak R, Bhat K, Somannavar P, Hosmani J. HSP70 induces TLR4 signaling in oral squamous cell carcinoma – an immunohistochemical study. J Can Res Ther. 2013; 9(4):624-9
- Nair S, Bhat K, Nayak R, Kotrashetti V, Babji D. Effect of Lactobacillus on Mutans Streptococcus in caries-free and high caries risk individuals. J Pharm Biomed Sci. 2013; 31(31): 1192-1198.


Kathie Tang is a member of the Terence Williams Lab at City of Hope.


Kevin Wang is a member of the Terence Williams Lab at City of Hope.


Linlin Yang, M.D., Ph.D., graduated from Binzhou Medical University China in 2005 with a bachelor’s degree in clinical medicine and then completed his M.Sc. in human anatomy and physiology at Southern Medical University China in 2008. He attained his Ph.D. in cancer biology in 2013 from the Fredrich-Schiller University Germany in the group of Professor Iver Petersen, working on the role of desmosome membrane proteins in human cancers.
Driven by “turning science from bench to clinic,” Dr. Yang joined in the lab of Terence Williams, Ph.D., group in 2014 to investigate the role of oncogene-mediated DNA damage repair in aggressive neoplasms, with a focus on KRAS mutations in pancreatic cancer, BRAF mutation in anaplastic thyroid cancer and SWI/SNF complex in lung cancer.
Research Focus
- Preclinical studies on testing chemoradiotherapy efficacy of small molecules
- Mechanisms of KRAS/BRAF mutation induced cancer treatment resistance
- Roles for SWI/SNF complex in tumorigenesis
Education
- 2009-2013, Dr.med. (summa cum laude), Friedrich Schiller University Jena, Germany
- 2005-2008, M.S., Human Anatomy and Physiology, Southern Medical University, China
- 2000-2005, B.S., Medicine, Binzhou Medical College, China
Internship
- 2004-2005, Intern, Internal Medicine, Binzhou Medical College Affiliated Zibo Center Hospital, China
Professional Experience
- 2020-present, Staff Scientist, Department of Radiation Oncology, City of Hope, Duarte, California, U.S.
- 2017-2020, Research Associate, Department of Radiation Oncology, OSUCCC, U.S.
- 2014-2017, Postdoctoral Researcher, Department of Radiation Oncology, OSUCCC, U.S.
- 2013-2013, Postdoctoral Researcher, Friedrich Schiller University, Jena, Germany
Academic and Professional Honors
- 2019, STT Travel Award, Radiation Research Society, U.S.
- 2018, Basic research seed grant, Department of Radiation Oncology, OSUCCC
- 2016, 3rd Place Poster Competition, OSUCCC Annual Scientific Meeting
- 2013, JIP Cancer Graduation Award 2013, Friedrich Schiller University, Jena, Germany
- 2013, Dissertation: summa cum laude, Friedrich Schiller University, Jena, Germany
- 2013, Young Investigator Award,22nd Asia Pacific Cancer Conference
- 2013, DAAD conference and lecture travel scholarship, Germany
- 2013, Chinese government award for outstanding self-financed students abroad
- 2010, Jena Interdisciplinary Center for Clinical research funding, Jena, Germany
Memberships
- American Association for Cancer Research
- Radiation Research Society
Representative Publications
- Yang L, Shen C, Estrada-Bernal A, et al. Oncogenic KRAS drives radioresistance through upregulation of NRF2-53BP1-mediated non-homologous end-joining repair. Nucleic Acid Res. 2021; 49(19):11067-11082
- Li Z, Yang L*, Wang H, et al. Non-Small-Cell Lung Cancer Regression by siRNA Delivered Through Exosomes That Display EGFR RNA Aptamer. Nucleic Acid Ther. 2021;31(5):364-374 (*co-first author)
- Shen C, Shyu DL, Xu M, Yang L, Webb A, Duan W, Williams TM. Deregulation of AKT-mTOR signaling contributes to chemoradiation resistance in lung squamous cell carcinoma. Mol Cancer Res. 2021 Nov 22:molcanres.0272.2021
- Wolfe AR, Robb R, Hegazi A, Abushahin L, Yang L, Shyu DL, Trevino JG, Cruz-Monserrate Z, Jacob JR, Palanichamy K, Chakravarti A, Williams TM. Altered Gemcitabine and Nab-paclitaxel Scheduling Improves Therapeutic Efficacy Compared with Standard Concurrent Treatment in Preclinical Models of Pancreatic Cancer. Clin Cancer Res. 2021;27(2):554-565
- Yang L, Shen C, Pettit CJ, et al. Wee1 Kinase Inhibitor AZD1775 Effectively Sensitizes Esophageal Cancer to Radiotherapy. Clin Cancer Res. 2020;26(14):3740-3750
- Yang L, Kumar B, Li TY, et al. LCL161, a SMAC mimetic, induces preferential radiosensitization in human papillomavirus negative head and neck squamous cell carcinoma through induction of apoptosis. Mol Cancer Ther. 2019;18(6):1025-1035.
- Robb R, Yang L, Shen C, Wolfe AR, Webb A, Zhang X, Vedaie M, Saji M, Jhiang S, Ringel MD, Williams TM. Inhibiting BRAF Oncogene-Mediated Radioresistance Effectively Radiosensitizes BRAFV600E-Mutant Thyroid Cancer Cells by Constraining DNA Double-Strand Break Repair. Clin Cancer Res. 2019;25(15):4749-4760.
- Yang L, Patrick Wald, Zuo L, et al. Significant and Durable Clinical Response to Sorafenib and Radiation Therapy for a Patient With Stage IV Hepatocellular Carcinoma and LRRK2 Mutation. JCO Precision Oncology 2019;3:PO.18.00277. doi: 10.1200/PO.18.00277.
- Wu X, Li S, Hu X, Xiang X, Halloran M, Yang L, Williams TM, Houghton, PJ, Shen C, He Z. mTOR Signaling Upregulates CDC6 and Promotes the Loading of DNA Replication Helicase. Sci Rep 2019, 9(1):9805.
- Estrada-Bernal A, Chatterjee M, Haque SJ, Yang L, et al. MEK Inhibitor GSK1120212-Mediated Radiosensitization of Pancreatic Cancer Cells Involves Inhibition of DNA Double-Strand Break Repair Pathways. Cell Cycle 2015;14(23):3713-24.
- Chen Y*, Yang L*, Cui T, et al. The homeobox gene HOPX acts as a tumor suppressor by induction of cellular senescence in human lung cancer. J Pathol. 2015;235(3):397-407. (*co-first author)
- Yang L, Chen Y, Cui T, et al. Desmoplakin acts as a tumor suppressor by inhibition of the Wnt/b-Catenin signaling pathway in human lung cancer. Carcinogenesis 2012;33(10):1863-70
- Yang L, Chen Y, Cui T., et al. Identification of biomarkers to distinguish clear cell sarcoma from malignant melanoma. Hum Pathol. 2012;43(9):1463-70
- Cui T, Chen Y, Yang L, et al. The p53 target gene desmocollin 3 acts as a novel tumor suppressor through inhibiting EGFR/ERK pathway in human lung cancer. Carcinogenesis 2012;33(12):2326-33
- Cui T, Chen Y, Yang L, et al. DSC3 expression is regulated by p53, and methylation of DSC3 DNA is a prognostic marker in human colorectal cancer. Br J Cancer. 2011;104(6):1013-9
- Cui T, Chen Y, Knösel T, Yang L, et al. Human complement factor H is a novel diagnostic marker for lung adenocarcinoma. Int J Oncol. 2011; 39, 161-8
- Chen Y, Cui T, Yang L, et al. The diagnostic value of cytokeratin 5/6, 14, 17, and 18 expression in human non-small cell lung cancer. Oncology 2011;80(5-6):333-40
- Chen Y, Cui T, Knösel T, Yang L, Zöller K, Petersen I IGFBP7 is a p53 target gene inactivated in human lung cancer by DNA hypermethylation. Lung Cancer 2011;73(1):38-44.


Aaron Zhaos is a member of the Terence Williams Lab at City of Hope.
- Adam Wolfe M.D., Ph.D.
- Nikhil Sebastian, M.D.
- Eric Miller, M.D., Ph.D.
- Moumita Chatterjee, Ph.D.
- Adriana Estrada, Ph.D.
- Longzhu Piao, Ph.D.
- Sooin Bae, Ph.D.
- Ryan Robb, B.S.
- Rebecca Packard, B.S.
- Andrew Hu, B.S.
- Ahmad Hegazi, B.S.
- Megan Halloran, B.S.
- Duan-Liang “Bruce” Shyu, M.S.
- Marally Vedaie, B.S.
- C. Andrew Robinson, B.S.
- Chen Lin, B.S.
- David Morrell, B.S.
- Mitchell Romito, B.S.
- Michael Lause, B.S.
- Matthew Harbrecht, B.S.
- Joseph Salloum, B.S.
- Tianyun “Ardy” Li
- Cory Pettit, B.S.
- Steve Walston, M.D.
- Patrick Wald, M.D.
- Vaibhava Kesarwani
- Eric Perez, B.S.
We collaborate with organizations in progressing the development of new treatments in our specialized areas of research.

- Yang L, Shen S, Estrada-Bernal A, Robb R, Chatterjee M, Sebastian N, Webb A, Mo X, Chen W, Krishnan S, Williams TM. Oncogenic KRAS drives radioresistance through upregulation of Nrf2-53BP1-mediated non-homologous end-joining repair. Nucleic Acids Research. 2021 Nov 8;49(19):11067-11082. PMID: 34606602; PMCID: PMC8565339.
- Wolfe AR, Chablani P, Siedow MR, Miller ED, Walston S, Kendra KL, Wuthrick E, Williams TM. BRAF Mutation Correlates with Worse Local-regional Control Following Radiation Therapy in Patients with Stage III Melanoma. Radiation Oncology. 2021 Sep 18;16(1):181. PMID: 34537078; PMCID: PMC8449455.
- Robb R, Kuo JC, Liu Y, Corrales-Guerrero S, Cui T, Hegazi A, Nagy G, Lee RJ, Williams TM. A novel protein-drug conjugate, SSH20, demonstrates significant efficacy in caveolin-1-expressing tumors. Molecular Therapy Oncolytics, 22(24): 555-564. PMID: 34553040. PMCID: PMC8433067.
- Sebastian NT, Webb A, Merrell KW, Koay EJ, Wolfe AR, Zhang L, Wilhite TJ, Elganainy D, Robb R, Chen W, Cloyd J, Dillhoff M, Tsung A, Abushahin L, Noonan A, Williams TM. Development of a MicroRNA Signature Predictive of Recurrence and Survival in Pancreatic Ductal Adenocarcinoma. Cancers (Basel). 2021 Oct 15;13(20):5168. PMID: 34680317; PMCID: PMC8534163.
- Shen C, Shyu DL, Xu M, Yang L, Webb A, Duan W, Williams TM. De-regulation of AKT-mTOR signaling contributes to chemoradiation resistance in lung squamous cell carcinoma. Mol Cancer Res. 2022 Mar 1; 20(3):425-433. doi: 10.1158/1541-7786.MCR-21-0272. PMID: 34810212 PMCID: PMC8898295.
Latest Research News
34.1293487, -117.9726643
Duarte, CA 91010


