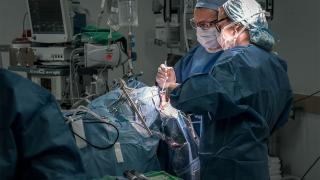
This is a vivid account of a surgical procedure performed at City of Hope. The details may be disturbing for some readers.
Striding through the swinging doors that lead into the operating theater, his arms upright and dripping water after scrubbing in, Behnam Badie, M.D., carries himself with the sure air of a neurosurgeon.
The procedure he is about to perform, involving meticulously excavating a tumor from a patient's brain, is one he has done thousands of times. But there is one difference.
If the patient, a 44-year-old male, has active tumor growth, he will be eligible for a clinical trial involving using his own immune system cells to fight his brain cancer.
This patient has had a glioblastoma, which is a pretty aggressive tumor, says Badie, chief of the Division of Neurosurgery at City of Hope, as a nurse wraps him in a gown before surgery. "Our plan is to explore the previous surgical area, expose the brain and get biopsies. If there's evidence that there's recurrence, the plan is to remove the tumor."
For this and other patients being considered for the clinical trial, qualifying is a complicated reality. No new tumor growth is good news, but likely temporary. Glioblastomas almost always come back, quickly and with a vengeance.
On the other hand, even a shred of evidence of tumor growth for this patient would mean qualifying for the trial, and being treated with an advanced therapy that could extend his life.
Rescue Mission
The surgery is a rescue mission in two phases. The first, removing a tumor that is impairing the patient's physical functioning, and causing his moods, according to his wife, to swing wildly from listless to aggressive to confused. The second phase involves preparing the patient for a new type of therapy called chimeric antigen receptor, or CAR T cell therapy to target his brain tumor.
"We all know the three main pillars for cancer therapy: surgery, chemotherapy and radiation," says Christine Brown, Ph.D., associate director of the T Cell Therapeutics Research Laboratory at City of Hope and co-lead with Badie on the trial. "And we've improved in all those areas, but I think the most exciting is this new fourth pillar of immunotherapy where we're trying to empower a patient's immune system to both recognize and destroy their cancer.
"We're utilizing the power and potency of specialized white blood cells called T cells and reprogramming them. What this does is allow T cells to see cancer cells that they couldn't see before. And when they see them, they seek out those cells and kill them."
How CAR T cell therapy works, as explained by Saul Priceman, Ph.D.
What is unique about the therapy is that CAR T cells, "immune system cells, removed from the patient, genetically altered, then reintroduced to the patient to bolster his body's ability to fight his cancer” will be infused directly to the brain using delivery tubes called catheters.
One catheter will be pointed at the cavity where the patient's tumor was growing, to target any leftover cancer cells. The other will be directed into the cerebrospinal fluid, a clear liquid that bathes the brain and spinal cord, to do the same.
"This direct-to-brain approach is unique to City of Hope," says Brown, The Heritage Provider Network Professor in Immunotherapy. "We were the first to implement this approach. Very few centers in the world can do this."
This rescue mission has tremendous implications. The patient has three young children and a wife whose lives will be challenged if he is not enrolled in the clinical trial. A family's future hangs in the balance of this surgery's outcome.
11:32 A.M.
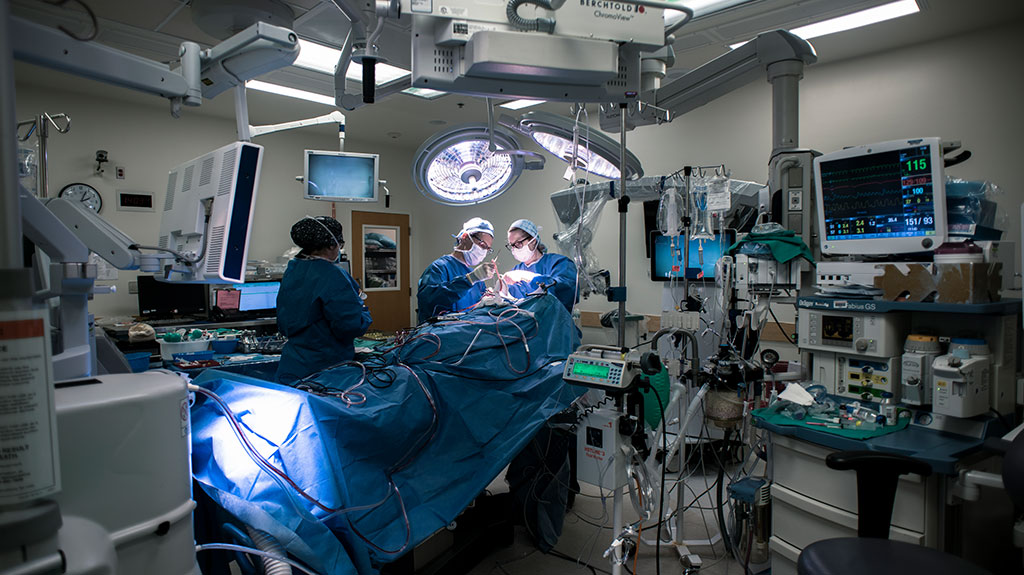
Two nurses and neurosurgeon Lisa Feldman, M.D., Ph.D., position the patient on the table so his head is angled properly for surgery. A nearby whiteboard, in shorthand, explains the procedure: "Right craniotomy for tumor resection. Placement of Rickham reservoir and catheter."
A screen above the operating table displays the silver contours of the patient's face alongside digital images of the interior of his brain. Feldman points a long metal navigation probe, which looks a little like an old-fashioned TV antenna, toward a section of the patient's skull. With each touch of the probe, a 3D image of a section of the brain is revealed on a nearby screen.
It is used by the surgeons throughout the procedure to guide them, indicating where they are in the brain, and which structures could be affected by their surgical instruments.
"It allows us to see how far we are in certain brain regions," says Badie. "To see whether we are almost at the expected edge of where the tumor is growing."
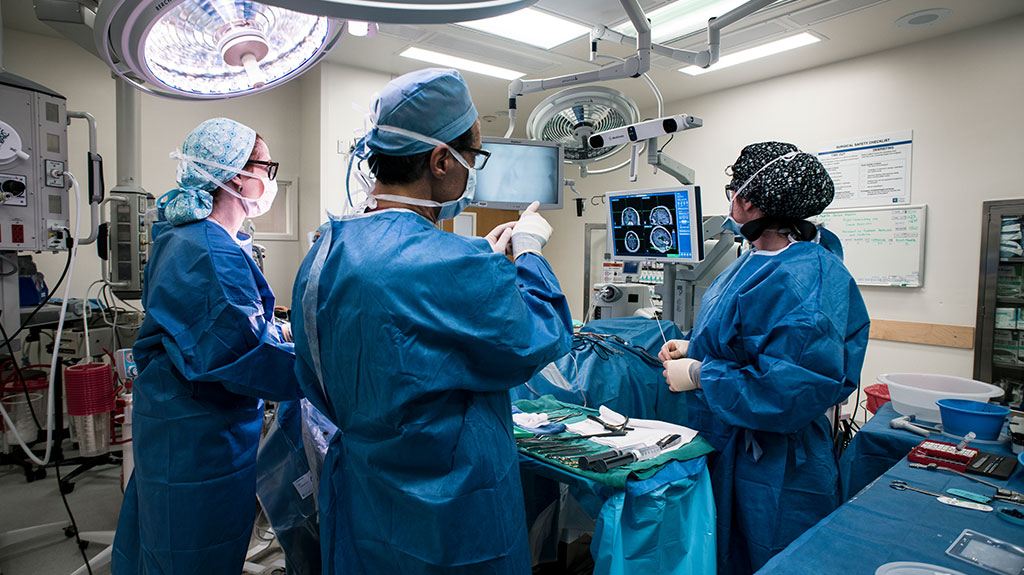
11:47 A.M.
"Incision," says Feldman as her voice rings through the OR. She begins to cut through the patient's scalp. Underneath is a bone flap, a section of the skull bone that will be removed, revealing the portal through which the surgeons will access his brain.
Badie joins Feldman and the two neurosurgeons hover over the patient, working together to remove tiny flecks of bone that surround the flap; putting them, one by one, into a plastic cup. Soon, they will use a screwdriver-type device to dislodge the bone.
An hour earlier, the patient, whose history includes three years of treatment, including brain surgeries, radiation, chemotherapy and experimental therapies, told his wife he wanted to give up. There had been too many surgeries. Too many procedures. Too much time trying to save his life at the expense of living out his remaining days.
"I told him, 'You have two options,'" recalls his wife. "'You go home, wait for the tumor to keep growing and you die. Or you go through another surgery and say to yourself, at least I tried it all.'"
12:00 P.M.
The bone flap removed, Badie points the navigation probe toward the brain. He begins cutting and peeling away the dura, a thick, protective layer of tissue that covers the brain and spinal cord. Soon, the patient's brain is exposed: a spongy mass of grayish-pink fatty tissue.
Badie extracts a piece of tissue for biopsy: "Right frontal glioma versus gliosis," he says, indicating the type of tissue to be assessed by pathology, either tumor tissue or scar tissue.
"Navigation probe," says Badie, gesturing again for the instrument that helps visualize the brain.
Badie then maneuvers a large microscope so it is positioned above the opening in the patient's brain.
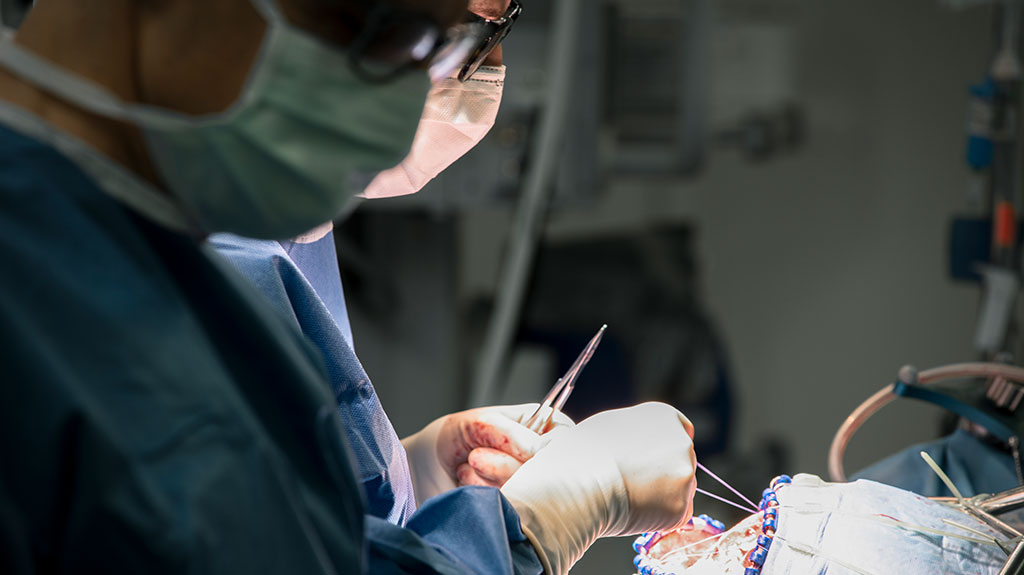
12:09 P.M.
The surgeons take turns navigating brain areas that a recent MRI indicated could be harboring new tumor growth, pulling out fatty white tissue as they go.
"Looks like he had a hemorrhage," says Feldman, as she holds up the next sliver of tissue, which has more of a brownish tinge.
Inside the OR there is a rhythm unfolding: the sound of suction to remove tiny rivulets of blood bubbling up from the brain, alternating with the beep of a cauterizing device. The patient's brain appears to be pulsating in response to the surgeons' work.
Feldman cranes her neck at intervals to get a better look at the navigation screen. She asks for a receptacle for a specimen, remarking that it "looks like scar tissue."
Badie begins calling out the type of specimen, "This is a..." before a nurse finishes his sentence, "right frontal glioma versus gliosis."
After handing over the specimen, Badie turns to Feldman and says, "We need to save a lot of that tissue."
Enough tissue to get an accurate gauge of whether there is active tumor growth.
12:19 P.M.
"That's scar," says Feldman, holding up brain tissue. "See it? I'll stay away from that."
She continues pulling at tiny popcorn-like parcels of tissue inside the patient's brain. A red light shines into a region that, magnified by the microscope, looks like a cavernous tunnel in the brain. Feldman uses a scraping motion as she digs, as if she is carving out a mini ditch around the tissue she is targeting.
"Get a little bit of the sides," says Badie. "Make sure you remove the tissue."
"Can I have the scope?" says Feldman.
12:26 P.M.
Feldman continues to pull out white slivers of scar tissue. She uses long tweezers to transfer each fatty looking piece to a tray, preparing to send them to pathology.
The challenge is that the scar tissue is in the resection cavity, the space where the patient's tumor has grown in the past. If it turns out the greyish hues they saw on the MRI are not new growth, but old scar tissue, the patient will not be eligible for the trial.
"What we could be seeing is radiation necrosis," says Feldman. "It's actually one of the biggest problems in neurosurgery. Glioblastoma is a nasty tumor, so we often don't know if what we're seeing is radiation injury from all the treatment the patient has undergone, or it's tumor coming back."
Inside brain surgery. WARNING: Some scenes in this video are graphic.
12:34 P.M.
"This part feels really firm," says Feldman, as she continues probing the brain. Meanwhile, Badie is on the phone with pathology, getting the analysis of tissue previously sent.
"The pathologist called back," says Badie. "He sees abnormal cells but he's not sure if it's new tumor growth or the previous tumor. So we are going to send some new samples to see if we have some viable tumor cells."
12:57 P.M.
Badie places a small mesh sheet into the tumor cavity to help stanch bleeding. He alternates between this and cauterizing, or burning, tissue to keep small blood vessels in the area from bleeding.
"If we don't have hemostasis, the patient could develop a blood clot in the surgical cavity," explains Badie.
Feldman is on the phone with pathology, getting final confirmation about the patient's eligibility for the trial.
"Okay, focally recurrent GBM with the background of necrosis," says Feldman to the pathologist before turning to Badie and saying, "We got our diagnosis."
"That's a bittersweet diagnosis," she continues. "Both good because now he's going to be eligible to get into the clinical trial, but obviously it's bad to have a recurrent tumor. But with this tumor it's expected."
A nurse places thin tubes and two small dome-shaped reservoirs on a tray so Badie and Feldman can assemble and later implant them.
Badie examines sections of tumor tissue he is excising and hands them to a nurse who drops them into tubes nearly filled with colored liquid.
1:21 P.M.
The image being transmitted by the overhead microscope reveals the path traveled by a probe being used to position one of the catheters. As it penetrates the brain tissue, bright reddish and gray hues splash across the screen. The inside of the patient's brain, during this part of the procedure, looks like a piece of abstract art.
Feldman uses the scope to help position a catheter so that it points toward the tumor cavity.
"Can I have the reservoir?" asks Feldman, gesturing for the device that will help deliver the CAR T cells while she again irrigates the wound opening.
"I'm getting the second catheter ready," says Badie, as he assembles the tubing and reservoir that will be placed near the cavity in the middle of the brain that houses much of the patient's cerebrospinal fluid.
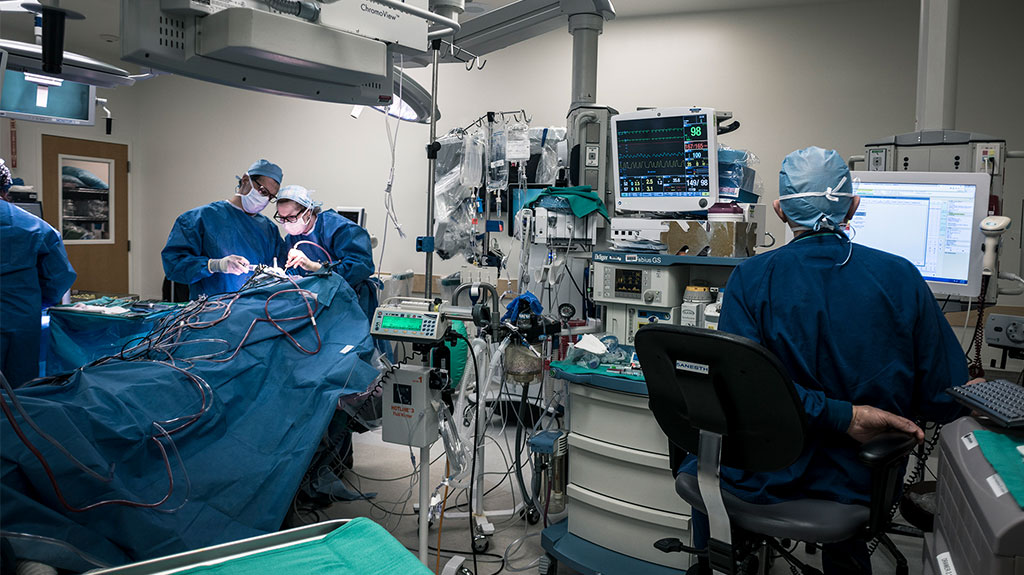
1:43 P.M.
As Badie places the second catheter, Feldman begins drilling tiny screws into the bone flap to prepare it to again cover the patient's brain.
An air of finality has settled into the operating room. The anesthesiologist checks the patient's stats. A nurse walks in with a kit that will be used to repair the patient's dura. Nearby, nurses work together to count sponges and equipment.
1:56 P.M.
The neurosurgeons stand at either side of the surgical wound and alternate between stitching the dura closed using thin, thread-like sutures.
The rescue mission was successful. The patient qualified for the trial.
"For us it is a mixed feeling," says Badie. "You don't want there to be tumor and you're hopeful it's just scar. So, it's sad this patient has recurrent disease. But at least we can try and improve his quality of life and longevity with CAR T cell therapy."