
City of Hope has announced that 23% of patients with relapsed or treatment-resistant KMT2Ar acute myeloid leukemia (AML), acute lymphoid leukemia (ALL) and mixed phenotype leukemia who took the small molecule drug revumenib in the AUGMENT-101 multicenter clinical trial had a complete remission (CR) (the cancer went into remission with complete recovery of peripheral blood counts) or a CRh (the cancer went into remission and there was partial recovery of peripheral blood counts). The data was presented as a late-breaking abstract at the American Society of Hematology (ASH) meeting in San Diego and in a press briefing on Dec. 11. Revumenib is a highly selective inhibitor of the menin-KMT2A binding interaction that is being developed by Syndax Pharmaceuticals.
The 13 patients with CR or CRh also stayed in remission for an average of 6.4 months, with six patients still remaining in remission at the time the data was collected. Trial participants had an overall response rate (ORR) of 63%, or 36 out of 57 patients, meaning they had a partial or complete clearance of the leukemia in response to the therapy with or without blood count recovery.
“The only current treatment plan for adult and pediatric patients with these types of relapsed or treatment-resistant leukemias is chemotherapy to achieve temporary remission as a path to a bone marrow or stem cell transplant. But fewer than 10% of these patients respond to the chemotherapy, and the expectation for survival is in the range of three months,” said Ibrahim Aldoss, M.D., City of Hope associate professor in the Division of Leukemia within the Department of Hematology & Hematopoietic Cell Transplantation and principal investigator for the AUGMENT-101 trial. “What this trial demonstrates is that revumenib did work safely and effectively in varying degrees in a majority of trial participants and had induced deep remissions in the majority of responders, giving us hope about the prospect of an additional, urgently needed treatment option, as well as a therapy that can help these patients to receive a curative bone marrow or stem cell transplant.”
Thirty-nine percent of patients who had ORR, or 14 total, were able to successfully receive a bone marrow or stem cell transplant — the only treatment that can cure these leukemia types — while their cancer was in remission with the therapy. Aldoss explained that this finding is important since patients with these leukemias cannot have a transplant if their bone marrow continues to have disease. In addition, half of transplanted patients took revumenib after transplant as a maintenance therapy to reduce the chance of their leukemia relapsing.
The study enrolled 92 patients with KMT2Ar leukemias, including children, adults and older adults, with various leukemia subtypes (AML, ALL and mixed phenotype leukemia). Fifty-seven patients in the largest group had their KMT2Ar finding confirmed by a central laboratory, and they were considered for the study’s efficacy endpoint. KMT2Ar is a specific genetic mutation sometimes found in the DNA of leukemia cells. It is characterized by chemotherapy resistance and a high relapse rate.
“We are thrilled to present additional detail on the positive results for revumenib in KMT2Ar acute leukemia that continue to demonstrate its consistently compelling clinical profile as a potential monotherapy for these heavily pretreated patients with high unmet need,” said Michael A. Metzger, chief executive officer of Syndax. “We look forward to submitting a new drug application to the U.S. Food and Drug Administration for revumenib for the treatment of R/R KMT2Ar acute leukemia by year-end, with the goal of efficiently bringing this treatment to patients who need it most.”
Aldoss noted that revumenib produced comparable response rates in patients of different ages and leukemia subtypes.
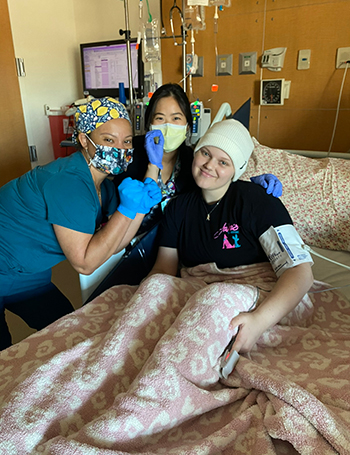
Tatum Demontmorency, 19, participated in the clinical trial at City of Hope after she was diagnosed with leukemia in 2021.The Bakersfield, California, resident began to feel weak and fatigued, and at first, doctors suspected a viral infection like mononucleosis. Demontmorency later started to experience severe stomach pain.
After more bloodwork and a computed tomography (CT) scan, she was diagnosed with AML with KMT2Ar. She was treated with standard chemotherapy, but unfortunately her leukemia returned later. Demontmorency and her family were advised to travel to Los Angeles for cancer treatment, and she eventually sought care at City of Hope’s Children’s Cancer Center, where she enrolled in the revumenib trial. The therapy, an oral pill that is taken twice a day in a 28-day cycle, cleared all her detectable cancer, and Demontmorency was able to receive a stem cell transplant from her brother, Trey.
Demontmorency is now a sophomore at Bakersfield College, has her eye on a career in health care, possibly as an ultrasound technician, and has returned to playing volleyball. She also coaches the volleyball team at her old high school.
"Tatum's story is truly inspirational, and our hope is that revumenib will continue to make it possible for our patients with these types of leukemias to continue with their lives,” Aldoss added.
Among trial participants, the therapy was safe and tolerable, with the most common side effects being nausea, differentiation syndrome and reversible changes in electrocardiogram readings. Patients tolerated these and other side effects well, as only 6% of patients left the trial due to treatment-related adverse effects.
If approved for use by the FDA, the product would be the first therapy to target what’s known as the menin-KMT2A interaction. Menin is a protein that binds to KMT2A, a genetic defect found in some leukemia cells, and this interaction causes KMT2Ar AML and ALL to accumulate abnormal cells in the bone marrow and blood. Revumenib can prevent this interaction from occurring, leading to possible cancer remission.
Among trial participants, the therapy was safe and tolerable, with the most common side effects of nausea, differentiation syndrome and reversible changes in electrocardiogram. Patients tolerated these and other side effects well as only 6% of patients left the trial due to treatment-related adverse effects.
If approved for use by the FDA, the product would be the first therapy to target what’s known as the menin-KMT2A interaction. Menin is a protein that binds to KMT2A, a genetic defect, and this interaction causes KMT2Ar AML and ALL to accumulate abnormal cells in the bone marrow and blood. Revumenib can prevent this interaction from occurring, leading to possible cancer remission.
At City of Hope Children’s Cancer Center, our expert providers are dedicated to delivering world-class, personalized care for children, adolescents and young adults. Each patient is cared for by a multidisciplinary team of oncologists, hematologists, surgeons, radiation therapists, pathologists and supportive care staff, including child life specialists. Our team has access to leading-edge treatments, including our world-renowned stem cell and bone marrow transplantation programs and the latest clinical trials. Our specialists work together to develop a unique, targeted treatment plan for every patient while providing family support. Continuing care into adulthood for survivors, our Childhood, Adolescent and Young Adult Survivorship Program provides specialized follow-up care for patients who have completed treatment for cancer or a similar illness that was diagnosed before 40 years of age.