A key member of the City of Hope team since 1996,
Amrita Krishnan, M.D., directs the Judy and Bernard Briskin Center for Multiple Myeloma Research at City of Hope, seeking to make gains in one of the most rapidly changing areas in cancer research. Dr. Krishnan serves as a steering committee member on the Multiple Myeloma Research Foundation and the North American representative to the International Myeloma Society, is the former chair of the Blood and Marrow Transplant Clinical Trials Network myeloma committee and is the co-chair of several of the largest myeloma transplant trials in the U.S.
City of Hope is always conducting groundbreaking clinical trials that look to advance the field of multiple myeloma treatment. Armed with a $4 million, multiyear R01 grant from the National Cancer Institute, we are now exploring the use of a 23-year-old rheumatoid arthritis drug, leflunomide, as a treatment for smoldering multiple myeloma (SMM), an early precursor to multiple myeloma. Research has already shown that leflunomide may help multiple myeloma patients by blocking enzymes needed for cell growth. Now, investigators want to see if leflunomide can also slow the precursor — delaying the progression of SMM into active myeloma, or even stopping it entirely.
“Leflunomide has a long safety record,” said multiple myeloma expert
Michael Rosenzweig, M.D., M.S., a co-principal investigator. “It’s been well-tested, it’s essentially nontoxic and well tolerated. Plus it’s much more affordable than any other multiple myeloma treatment.”
Given that SMM is twice as common in African Americans and has double the mortality rate among this population, conducting a thorough and diverse clinical trial is of utmost importance, and this is exactly what City of Hope plans to do.
“We’re going beyond statistical comparisons,” explained Kimlin Tam Ashing, Ph.D. “We’ll look at participants’ neighborhoods, their social structure, their experiences of discrimination, their ancestry” (a component to be handled by City of Hope’s ancestry expert, Rick Kittles, Ph.D., M.S.). “We’re going to examine the whole person. We want to understand people, including their response to treatment, beyond their diagnosis.”
In addition, City of Hope is also reporting promising results from a Phase 1 study conducted by Krishnan exploring the use of Talquetamab as an investigational therapy for the treatment of relapsed multiple myeloma. Talquetemab is a first-in-class bispecific IgG4 antibody that redirects T cells to kill multiple myeloma cells by binding to both GPRC5D and CD3 receptors. The findings indicate that Talquetamab is well tolerated and highly effective at the recommended Phase 2 doses.
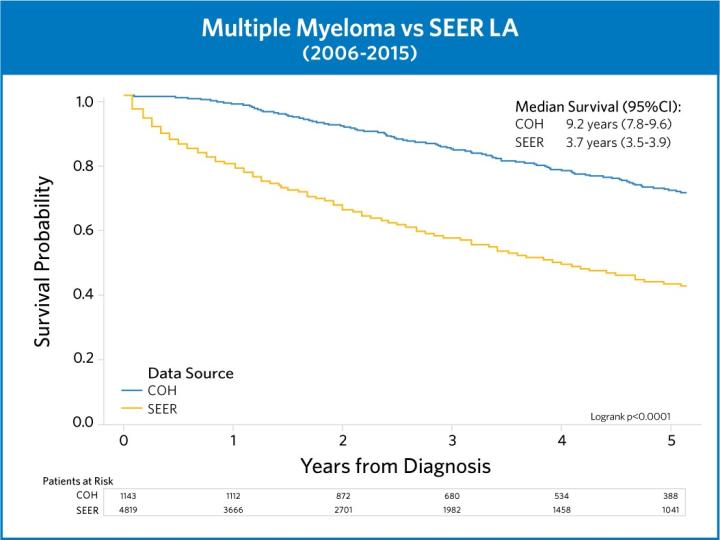
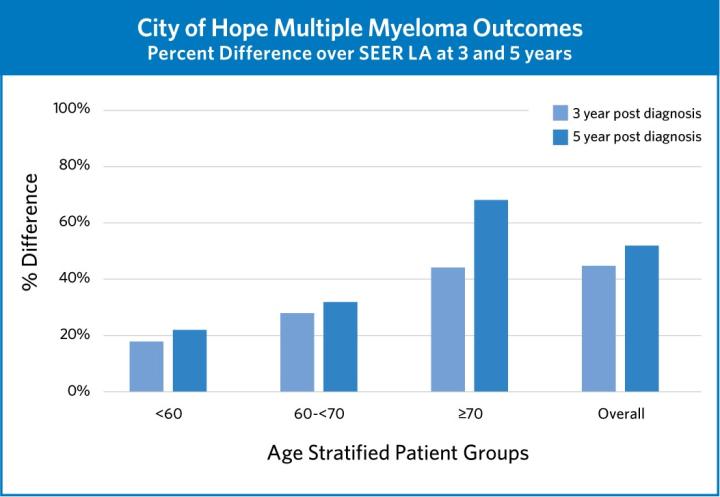
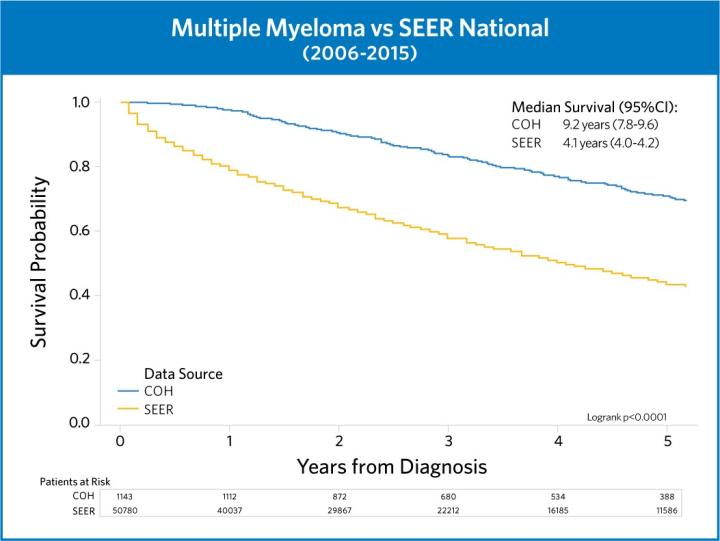
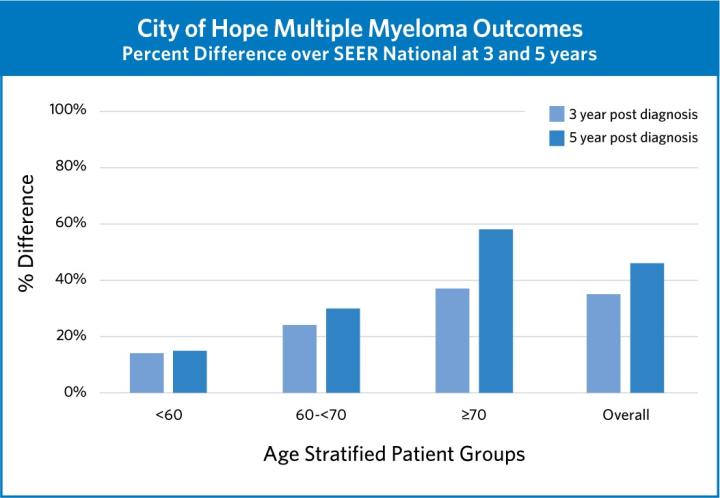
These trials and more are crucial to City of Hope’s success in achieving superior outcomes in myeloma treatment. By integrating the newest advances into our existing treatment paradigms, we are always confident that we are delivering the best possible care to our patients.