The Wanek Family Project for Type 1 Diabetes
In patients with type 1 diabetes, the insulin-producing cells in the pancreas, or beta cells, are destroyed by the immune system. The loss of beta cells leads to the inability to produce insulin and, in turn, control blood sugar levels, which results in life-threatening conditions if not treated. As insulin is essential for life, patients must be treated with insulin (using pumps or multiple daily injections). While the availability of human recombinant insulin (made possible by the seminal work of Arthur Riggs, Ph.D., at City of Hope®) represents a significant advance in treating and managing type 1 diabetes, it is not a cure.
The Wanek Family Project for Type 1 Diabetes at City of Hope® is a transformative partnership between a family committed to curing type 1 diabetes and City of Hope. Alberto Pugliese, M.D., joined City of Hope in late 2022 and serves as director of the project. He leads the team to apply a multifaceted approach to treating this complex and nuanced disease.
Our scientists are prioritizing developing combinatorial therapies that are considered essential to both prevent and reverse type 1 diabetes by:
Controlling autoimmunity by modulating the immune system to stop it from attacking and destroying the insulin-producing cells in the pancreas.
Correcting beta cell dysfunction by reversing the impaired function of insulin-producing beta cells in the pancreas and promoting their well-being.
Replacing lost beta cells by providing a sufficient number of insulin-producing cells to restore insulin production and reverse diabetes, noting that over time, most patients reach a stage when virtually all beta cells are lost.
Critically, City of Hope recognizes that actively collaborating with investigators and partners from other institutions is essential to accelerate progress and reach the goal of a cure for type 1 diabetes.
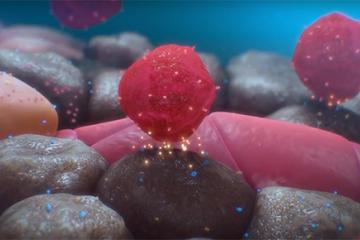
Because of our strength in advancing immunology and immunotherapies for other diseases, including cancer, City of Hope is a fascinating place to explore cell-based, immune-modulating approaches to type 1 diabetes. Our experts are advancing breakthrough interventions that correct autoimmunity by regulating — instead of suppressing — the immune system. Our innovative approaches aim to restore a balanced immune system to stop its attack on insulin-producing beta cells. Approaches include treatment with cell-based therapies that are manipulated to promote the regulation of the immune system and, in some cases, specific responses involved in destroying the pancreatic beta cells. Two such approaches are in clinical trials.
Renowned for our work in understanding precise features of the insulin-producing beta cells in health and diabetes, we are conducting vital research to correct and boost the function of insulin-producing cells, and some of our approaches can also reduce inflammation and stress of the beta cells. Approaches being developed include several new drugs that boost the function of insulin-producing cells and make them less susceptible to attack by the immune system and inflammation.

Over time, most patients reach a stage when virtually all beta cells are lost. Thus, reversing diabetes requires sufficient insulin-producing cells to restore sufficient and well-regulated insulin production. We are a national leader in islet cell transplantation to treat patients with diabetes, and we have an active trial enrolling patients. In addition, we are partnering in trials to advance therapies with stem-cell-derived insulin-producing beta cells. In parallel, our researchers are advancing compounds that promote the formation of new beta cells, which we aim to test in clinical trials in the future.
Since its inception, the Wanek Family Project has focused on developing and testing novel treatments for type 1 diabetes. These include three novel therapies in clinical trials or progressing toward a clinical trial.
TolDC trial: City of Hope is actively enrolling patients with type 1 diabetes in its Tolerogenic Dendritic Cell (TolDC) clinical trial to establish its safety and effectiveness. This immunotherapy involves administering modified immune cells to the patient to stop the attack on beta cells. Learn more about this promising study (NCT04590872).
CD6-CAR Treg trial: Through a synergistic collaboration, our diabetes and cancer scientists have developed a novel therapy with CD6-CAR Treg cells or modified regulatory T cells. As allowed by the FDA (Food and Drug Administration), we are initiating a clinical trial in patients with chronic graft-versus-host disease (GVHD); initial safety data from this trial will support proceeding with a clinical trial for patients with type 1 diabetes. Learn more about the CD6-CAR Treg cell therapy clinical trial (NCT05993611).
Fundamental work by Rupangi Vasavada, Ph.D., has led to the discovery that an existing drug used for another condition benefits the function of pancreatic beta cells. This has led to the organization of a clinical trial that will launch in 2024, with financial support from a JDRF grant.
Other examples of our innovative research:
- Borrowing a therapy initially developed for cancer patients, our scientists are developing a novel CD6/EGF Bionic drug to modulate the immune system and support insulin-producing cell health simultaneously. The drug is undergoing necessary testing before a clinical trial may be launched.
- We are developing a novel therapy that promotes the formation of new beta cells, which could restore insulin production by a patient's pancreas and potentially ameliorate or reverse diabetes. This therapy would benefit most type 1 diabetes patients, regardless of the stage of their disease. Read more about efforts to promote beta cell regeneration led by Adolfo Garcia-Ocaña, Ph.D., professor and chair of the Department of Molecular & Cellular Endocrinology and the Ruth B. & Robert K. Lanman Endowed Chair in Gene Regulation & Drug Discovery Research.
Diabetes Clinical Trials
Additional diabetes clinical trials are ongoing at City of Hope. Learn more about our diabetes clinical trials.
